Radiocarbon (Carbon-14) Dating: What It Is, Why It Works, and Where It Fails
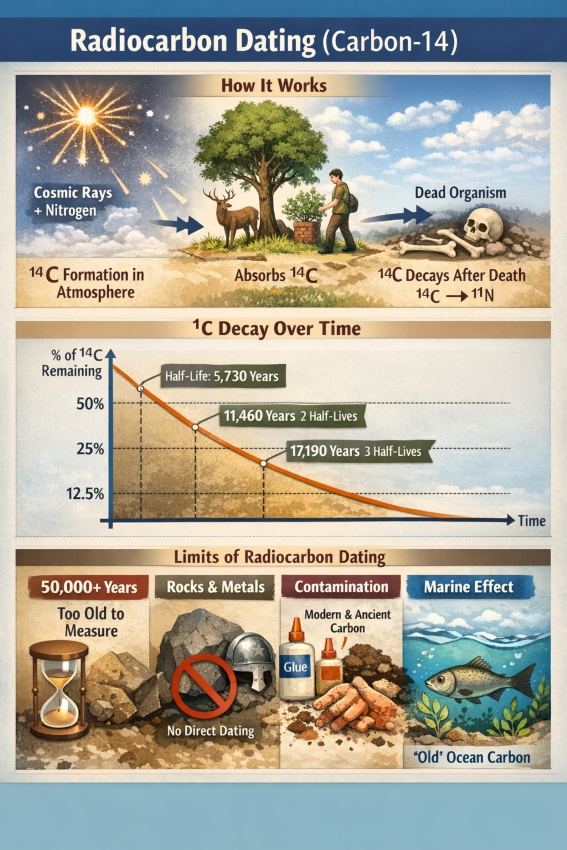
Radiocarbon dating—often called the carbon-14 (¹⁴C) method—is a technique used to estimate the age of once-living materials (wood, charcoal, bone, seeds, textiles, paper, etc.). It works by measuring how much radioactive carbon-14 remains in a sample compared with stable carbon, and using the known decay rate of ¹⁴C to calculate how long it has been since the organism died.
1) What is Carbon-14 (¹⁴C)?
Carbon exists in several forms called isotopes:
- ¹²C and ¹³C are stable (they do not decay).
- ¹⁴C is radioactive (it decays over time).
¹⁴C is continuously produced high in the atmosphere when cosmic rays interact with nitrogen. That newly formed ¹⁴C becomes part of atmospheric CO₂, and from there it enters the food chain.
2) Why radiocarbon dating works
The key idea: “Living things exchange carbon; dead things don’t.”
While an organism is alive, it constantly takes in carbon:
- Plants absorb CO₂ during photosynthesis.
- Animals eat plants and other animals.
Because of this ongoing exchange, living organisms tend to maintain a roughly steady ratio of ¹⁴C to stable carbon (¹²C/¹³C) that reflects the atmosphere (or their environment).
When the organism dies, carbon exchange stops. From that point onward:
- No new ¹⁴C is added.
- The existing ¹⁴C begins to decay back to nitrogen (¹⁴N).
So the amount of ¹⁴C steadily decreases relative to stable carbon, and that decrease acts like a clock.
3) The decay clock: half-life and age
¹⁴C decays at a predictable rate described by its half-life—the time it takes for half of the ¹⁴C atoms to decay.
- Half-life of ¹⁴C: ~5,730 years
If you measure that a sample has:
- 50% of the expected ¹⁴C → about 1 half-life → ~5,730 years old
- 25% → 2 half-lives → ~11,460 years old
- 12.5% → 3 half-lives → ~17,190 years old
Modern labs often use AMS (Accelerator Mass Spectrometry), which counts ¹⁴C atoms directly and can work with very small samples.
4) “Radiocarbon years” vs calendar years: calibration
A crucial nuance is that the amount of ¹⁴C in the atmosphere has not been perfectly constant over time. It varies due to factors such as:
- solar activity,
- Earth’s magnetic field strength,
- carbon cycle changes,
- and, in modern times, human activity.
Because of this, radiocarbon results are typically calibrated using independently dated records (especially tree rings, but also corals, lake sediments, etc.). Calibration converts a raw radiocarbon measurement into a calendar age range.
Practical takeaway: radiocarbon dating usually produces a date range, not a single exact year.
5) What radiocarbon dating can date well
Radiocarbon dating is best for once-living materials, for example:
- charcoal and wood
- seeds, grains, plant fibers
- bone collagen (the organic component)
- peat and soils rich in organic matter
- paper, parchment, textiles
- shell and coral (with important caveats—see limitations)
It is widely used in archaeology, paleontology, paleoclimate studies, and forensic/heritage science.
6) Major limitations and common pitfalls
Limitation A: It does not date most rocks or metals directly
Radiocarbon dating requires carbon that was part of a living system.
- Cannot date: igneous rocks, most minerals, metals, ceramics (in general)
- Can sometimes date indirectly: organic residue on pottery, charcoal in a sediment layer, wooden tools near a rock feature, etc.
Limitation B: Effective time range (too old / too young)
Because ¹⁴C keeps decaying, eventually so little remains that it becomes difficult to measure reliably.
- Typical practical upper limit: ~50,000–60,000 years
- For very recent materials, interpretation can also be complicated (see “bomb carbon” below).
Limitation C: Contamination can dominate the result
Radiocarbon dating is extremely sensitive to carbon that does not belong to the original sample.
Examples:
- modern carbon contamination (handling, conservation materials, glues)
- older carbon contamination (e.g., fossil carbon, carbonates, “dead carbon”)
Even a small amount of contamination can shift the age significantly, especially for small or very old samples. This is why sample cleaning and preparation are critical.
Limitation D: Reservoir effects (marine and freshwater)
Not all environments have the same effective ¹⁴C level as the atmosphere.
- Marine reservoir effect: ocean water can contain “older” carbon due to slow mixing with deep waters. Marine organisms can appear artificially old unless corrected.
- Freshwater reservoir / hardwater effect: lakes and rivers can carry dissolved ancient carbon from limestone, making fish, shells, or aquatic plants look older than they really are.
Limitation E: The “old wood” problem
Radiocarbon dates the time the carbon stopped exchanging—often the death of the organism, not the human event you care about.
Example:
- Dating a wooden beam tells you when the tree ring formed and the wood stopped exchanging carbon—not necessarily when the building was constructed.
- Reused timber can produce dates that are older than the structure.
Limitation F: Post-1950 complexity (“bomb carbon”)
Atmospheric nuclear testing in the 1950s–1960s greatly increased ¹⁴C levels (“bomb pulse”), which can complicate conventional interpretation.
Because of this, radiocarbon reporting uses a standard reference point:
- “Present” = 1950 CE
So many results are expressed as years BP (Before Present), meaning before 1950.
7) What a radiocarbon result really means
A strong radiocarbon interpretation typically includes:
- a measured radiocarbon age and uncertainty,
- calibration to calendar years (often as a probability distribution),
- contextual information (archaeological layer, associated materials),
- discussion of potential reservoir effects and contamination risks.
Radiocarbon dating is powerful, but it is not a magic timestamp—it is a measurement that becomes most reliable when combined with good context and careful lab practice.
Summary
Radiocarbon dating works because living organisms continuously take in ¹⁴C, but after death the ¹⁴C decays at a known rate. By measuring the remaining ¹⁴C and calibrating for historical atmospheric variation, scientists can estimate when an organism died.
It is highly effective for organic, once-living materials up to roughly 50,000–60,000 years, but it has important limitations: it does not date most rocks/metals directly, it can be distorted by contamination, it requires calibration, and it can be biased by reservoir effects and “old wood” scenarios.